Mothers were sent into a panic recently when various sources across social media claimed that Panadol Baby and Infant was to be suspended.
The alarm turned out to be nothing more than a rumour that got out of control suggesting the pain relief product was not safe.
With parents understandably concerned, the UAE Ministry was forced to step in and put the rumour right, revealing the information to be incorrect.
In a statement to the state news agency, Wam, the ministry urged the public to not publish any information from unofficial sources because “such information is, in most cases, imprecise and tendentious”.
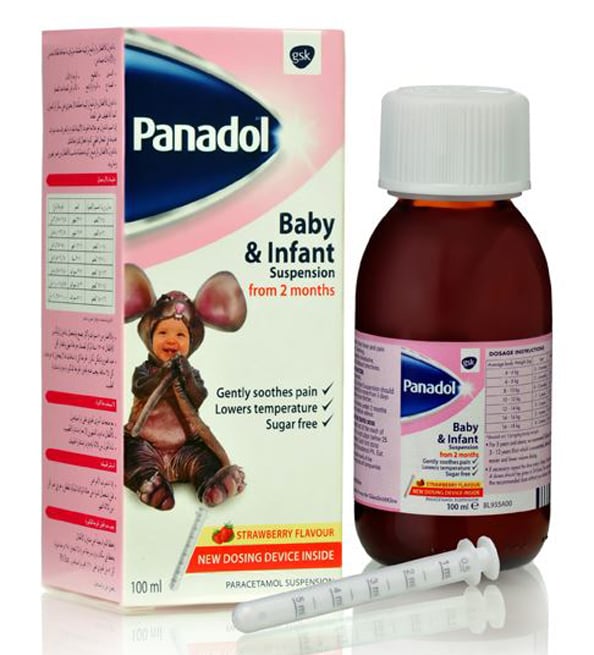
Rumour spread saying Panadol Baby and Infant had been suspended in the UAE
Amin Hussain Al Amiri, Under-Secretary of Public Health Policy and Licensing at the Ministry of Health and Prevention, and chairman of the Supreme Committee for Pharmaceutical Vigilance, noted that the incorrect and information and warnings about medical products were published by fear-mongering and bad-intentioned people. This, he added, could mislead patients into stopping to use prescribed medicines and exposing them to serious health risks.
Read: Spreading A Rumour Can Land You In Prison In The UAE
Read: Health Scares In The UAE You Should Know About
Read: 5 Key First Aid Tips For Parents
It is not known who started the rumour, and it has not been revealed whether the ministry will try to find the culprit.
Only last week we reported how anyone caught spreading a rumour through social media networks can now face up to three years in prison.
In fact the UAE’s Interior Ministry even set up a WhatsApp number in which worried citizens can report a rumour. There’s also a rumours page on Dubai Municipality website where you can check up whether something is true or false.
If you have any concerns about the safety of health and medical products in the UAE and any relevant announcements, Amiri has urged the public to contact the ministry via the following email address: pv@moh.gov.ae or via this hyperlink.
Image: Getty